Ethylene Glycol
| Use attributes for filter ! | |
| Boiling point | 197. 6 °C |
|---|---|
| Molar mass | 62. 07 g/mol |
| Formula | C2H6O2 |
| Density | 1. 11 g/cm³ |
| Melting point | -12. 9 °C |
| IUPAC ID | Ethane-1,2-diol |
| Date of Reg. | |
| Date of Upd. | |
| ID | 1212678 |
About Ethylene Glycol
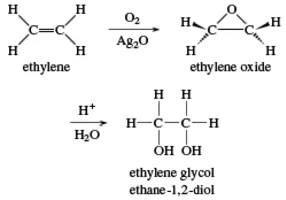
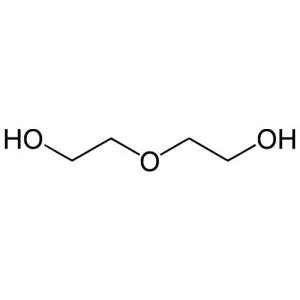
Ethylene glycol is an organic compound with the formula (CH₂OH)₂. It is mainly used for two purposes, as a raw material in the manufacture of polyester fibers and for antifreeze formulations. It is an odorless, colorless, sweet-tasting, viscous liquid. Ethylene glycol is toxic.
Boss of Indonesia cough syrup maker jailed after child deaths

... These batches contained 96% to 99% Ethylene Glycol, the prosecutor said...
Marion Biotech: Uzbekistan links child deaths to India cough syrup

... The ministry said that preliminary tests showed a batch of the medicine contained Ethylene Glycol, a toxic substance...
Cough-syrup scandal: How did it end up in The Gambia?

... Laboratory analysis of the syrups " confirms that they contain unacceptable amounts of diEthylene Glycol and Ethylene Glycol as contaminants" ...
WHO alert over India-made cough syrups after deaths in The Gambia

... The WHO said that laboratory analysis of samples of the products " confirms that they contain unacceptable amounts of diEthylene Glycol and Ethylene Glycol as contaminants...
Marion Biotech: Uzbekistan links child deaths to India cough syrup
Uzbekistan's health ministry has said that 18 Children have died after drinking a cough syrup manufactured by Indian drug maker Marion Biotech.
The ministry said that preliminary tests showed a batch of The Medicine contained Ethylene Glycol , a toxic substance.
The Children were given the Dok-1 Max syrup without a doctor's prescription, it said.
The amount they consumed also exceeded The Standard dose for Children .
The allegation from Uzbekistan comes weeks after to cough syrups made by another Indian firm.
India's health ministry and Marion Biotech have not responded to The Bbc 's request for comment yet. cited a government source as saying that " the health ministry was looking into the matter".
News agency ANI a Marion Biotech executive as saying that The Company has halted production of the syrup temporarily. He added that the government was conducting an enquiry and that The Firm would take action accordingly.
Marion Biotech is based in Noida, near India's national capital Delhi. Its website is currently down, but The Company 's LinkedIn page says it was founded in 1999 and that its products are " household names in Central Asian countries, Central and Latin America , South East Asia and Africa".
India produces a third of The World 's medicines, mostly in the form of generic drugs.
The country, home to some of the fastest-growing pharmaceutical companies, is known as the " world's pharmacy" and meets much of the medical needs of developing countries.
The Uzbek ministry statement, dated 27 December, says that Dok-1 Max tablets and syrup have been sold in the country since 2012.
" It was found that The Deceased Children , before admission to hospital treatment, took this drug At Home for 2-7 days, 3-4 times A Day , 2. 5-5ml, which exceeds The Standard dose of the drug for Children , " the ministry said.
The Statement did not specify over what time period the deaths occurred. BBC Monitoring had reported on 23 December, citing news website Gazeta. uz, that Uzbek authorities were investigating " claims that 15 Children died in central Samarkand region over The Past two months after taking a cough syrup made in India".
On 26 December, Podrobno. uz news website reported that 21 Children - 15 of them under the Age Of Three - were treated for acute kidney failure " allegedly caused by the India-made cough syrup Dok-1 Max between September and December". Three of the patients recovered.
The ministry also said that " preliminary laboratory studies have shown that this series of Dok-1 Max syrup contains Ethylene Glycol ".
In October, The World Health Organization (WHO) had and linked four India-made cough syrups to the deaths of 66 Children from kidney injuries in The Gambia . It said tests on samples of the syrup showed that they contained unacceptable amounts of toxic substances Diethylene Glycol and Ethylene Glycol .
Both The Indian government and The Company , Maiden Pharmaceuticals, have denied the allegations.
India that tests on The Four syrups showed that they complied with specifications, and a government official told The Bbc that The Who had been " presumptuous" in blaming the syrups. But the Who Said it stood by The Action taken.
Last week, a parliamentary committee in The Gambia of Maiden Pharmaceuticals after weeks of investigation. The Committee also recommended banning all products by The Firm in the country.
Read more India stories from The Bbc :Source of news: bbc.com