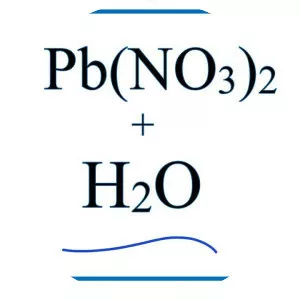Zinc Nitrate
| Use attributes for filter ! | |
| Formula | Zn(NO₃)₂ |
|---|---|
| Molar mass | 189. 36 g/mol |
| Solubility | alcohol |
| Solubility in water | 327 g/100 mL, 40 °C (trihydrate); 184. 3 g/100 ml, 20 °C (hexahydrate) |
| Boiling point | ~ 125 °C (257 °F; 398 K) decomposes (hexahydrate) |
| UN number | 1514 |
| Date of Reg. | |
| Date of Upd. | |
| ID | 2472270 |
About Zinc Nitrate
Zinc nitrate is an inorganic chemical compound with the formula Zn(NO₃)₂. This white, crystalline solid is highly deliquescent and is typically encountered as a hexahydrate Zn(NO₃)₂•6H₂O. It is soluble in both water and alcohol.